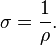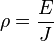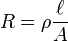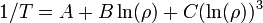Electrical resistivity (also known as resistivity, specific electrical resistance, or volume resistivity) is a measure of how strongly a material opposes the flow of electric current. A low resistivity indicates a material that readily allows the movement of electric charge. The SI unit of electrical resistivity is the ohm metre (Ωm). It is commonly represented by the Greek letter ρ (rho).
Electrical conductivity or specific conductance is the reciprocal quantity, and measures a material’s ability to conduct an electric current. It is commonly represented by the Greek letter σ (sigma), but κ (esp. in electrical engineering) or γ are also occasionally used. Its SI unit is siemens per metre (S·m−1) and CGSE unit is reciprocal second (s−1):
Contents |
Definitions
Electrical resistivity ρ (Greek: rho) is defined by,
where
- ρ is the static resistivity (measured in ohm-metres, Ω-m)
- E is the magnitude of the electric field (measured in volts per metre, V/m);
- J is the magnitude of the current density (measured in amperes per square metre, A/m²).
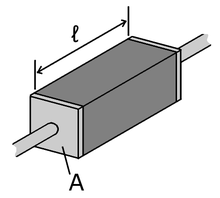
Many resistors and conductors have a uniform cross section with a uniform flow of electric current and are made of one material. (See the diagram to the right.) In this case, the above definition of ρ leads to:
where
- R is the electrical resistance of a uniform specimen of the material (measured in ohms, Ω)
 is the length of the piece of material (measured in metres, m)
is the length of the piece of material (measured in metres, m)- A is the cross-sectional area of the specimen (measured in square metres, m²).
The reason resistivity has the dimension units of ohm-metres can be seen by transposing the definition to make resistance the subject:
The resistance of a given sample will increase with the length, but decrease with greater cross-sectional area. Resistance is measured in ohms. Length over area has units of 1/distance. To end up with ohms, resistivity must be in the units of “ohms × distance” (SI ohm-metre, US ohm-inch).
In a hydraulic analogy, increasing the cross-sectional area of a pipe reduces its resistance to flow, and increasing the length increases resistance to flow (and pressure drop for a given flow).
Resistivity of various materials
- A conductor such as a metal has high conductivity and a low resistivity.
- An insulator like glass has low conductivity and a high resistivity.
- The conductivity of a semiconductor is generally intermediate, but varies widely under different conditions, such as exposure of the material to electric fields or specific frequencies of light, and, most important, with temperature and composition of the semiconductor material.
The degree of doping in semiconductors makes a large difference in conductivity. To a point, more doping leads to higher conductivity. The conductivity of a solution of water is highly dependent on its concentration of dissolved salts, and other chemical species that ionize in the solution. Electrical conductivity of water samples is used as an indicator of how salt-free, ion-free, or impurity-free the sample is; the purer the water, the lower the conductivity (the higher the resistivity). Conductivity measurements in water are often reported as specific conductance, relative to the conductivity of pure water at 25 °C. An EC meter is normally used to measure conductivity in a solution.
This table shows the resistivity, conductivity and temperature coefficient of various materials at 20 °C (68 °F)
| Material | ρ [Ω·m] at 20 °C | σ [S/m] at 20 °C | Temperature
coefficient[note 1] [K−1] |
Reference |
|---|---|---|---|---|
| Silver | 1.59×10−8 | 6.30×107 | 0.0038 | [1][2] |
| Copper | 1.68×10−8 | 5.96×107 | 0.0039 | [2] |
| Annealed Copper[note 2] | 1.72×10-8 | 5.80×107 | [citation needed] | |
| Gold[note 3] | 2.44×10−8 | 4.52×107 | 0.0034 | [1] |
| Aluminium[note 4] | 2.82×10−8 | 3.5×107 | 0.0039 | [1] |
| Calcium | 3.36×10−8 | 2.98×107 | 0.0041 | |
| Tungsten | 5.60×10−8 | 1.79×107 | 0.0045 | [1] |
| Zinc | 5.90×10−8 | 1.69×107 | 0.0037 | [3] |
| Nickel | 6.99×10−8 | 1.43×107 | 0.006 | |
| Lithium | 9.28×10−8 | 1.08×107 | 0.006 | |
| Iron | 1.0×10−7 | 1.00×107 | 0.005 | [1] |
| Platinum | 1.06×10−7 | 9.43×106 | 0.00392 | [1] |
| Tin | 1.09×10−7 | 9.17×106 | 0.0045 | |
| Lead | 2.2×10−7 | 4.55×106 | 0.0039 | [1] |
| Titanium | 4.20×10−7 | 2.38×106 | X | |
| Manganin | 4.82×10−7 | 2.07×106 | 0.000002 | [4] |
| Constantan | 4.9×10−7 | 2.04×106 | 0.000008 | [5] |
| Stainless steel[note 5] | 6.897×10−7 | 1.450×106 | [6] | |
| Mercury | 9.8×10−7 | 1.02×106 | 0.0009 | [4] |
| Nichrome[note 6] | 1.10×10−6 | 9.09×105 | 0.0004 | [1] |
| Carbon (amorphous) | 5 to 8×10−4 | 1.25 to 2×103 | −0.0005 | [1][7] |
| Carbon (graphite)[note 7] | 2.5 to 5.0×10−6 ⊥basal plane
3.0×10−3 //basal plane |
2 to 3×105 ⊥basal plane
3.3×102 //basal plane |
[8] | |
| Carbon (diamond)[note 8] | ~1012 | ~10-13 | [9] | |
| Germanium[note 8] | 4.6×10−1 | 2.17 | −0.048 | [1][2] |
| Sea water[note 9] | 2×10−1 | 4.8 | [10] | |
| Drinking water[note 10] | 2×101 to 2×103 | 5×10-4 to 5×10-2 | [citation needed] | |
| Deionized water[note 11] | 1.8×105 | 5.5 × 10−6 | [11] | |
| Silicon[note 8] | 6.40×102 | 1.56×10-3 | −0.075 | [1] |
| GaAs | 5×107 to 10-3 | 5×10-8 to 103 | [12] | |
| Glass | 1010 to 1014 | 10-11 to 10-15 | ? | [1][2] |
| Hard rubber | 1013 | 10-14 | ? | [1] |
| Sulfur | 1015 | 10-16 | ? | [1] |
| Air | 1.3×1016 to 3.3×1016 | 3 to 8 × 10−15 | [13] | |
| Paraffin | 1017 | 10-18 | ? | |
| Quartz (fused) | 7.5×1017 | 1.3×10-18 | ? | [1] |
| PET | 1020 | 10-21 | ? | |
| Teflon | 1022 to 1024 | 10-25 to 10-23 | ? |
The effective temperature coefficient varies with temperature and purity level of the material. The 20 °C value is only an approximation when used at other temperatures. For example, the coefficient becomes lower at higher temperatures for copper, and the value 0.00427 is commonly specified at 0 °C. For further reading: http://library.bldrdoc.gov/docs/nbshb100.pdf.
The extremely low resistivity (high conductivity) of silver is characteristic of metals. George Gamow tidily summed up the nature of the metals’ dealings with electrons in his science-popularizing book, One, Two, Three…Infinity (1947): “The metallic substances differ from all other materials by the fact that the outer shells of their atoms are bound rather loosely, and often let one of their electrons go free. Thus the interior of a metal is filled up with a large number of unattached electrons that travel aimlessly around like a crowd of displaced persons. When a metal wire is subjected to electric force applied on its opposite ends, these free electrons rush in the direction of the force, thus forming what we call an electric current.” More technically, the free electron model gives a basic description of electron flow in metals.
Temperature dependence
In general, electrical resistivity of metals increases with temperature, while the resistivity of intrinsic semiconductors decreases with increasing temperature. In both cases, electron–phonon interactions can play a key role. At high temperatures, the resistance of a metal increases linearly with temperature. As the temperature of a metal is reduced, the temperature dependence of resistivity follows a power law function of temperature. Mathematically the temperature dependence of the resistivity ρ of a metal is given by the Bloch–Grüneisen formula:
where ρ(0) is the residual resistivity due to defect scattering, A is a constant that depends on the velocity of electrons at the Fermi surface, the Debye radius and the number density of electrons in the metal. ΘR is the Debye temperature as obtained from resistivity measurements and matches very closely with the values of Debye temperature obtained from specific heat measurements. n is an integer that depends upon the nature of interaction:
- n=5 implies that the resistance is due to scattering of electrons by phonons (as it is for simple metals)
- n=3 implies that the resistance is due to s-d electron scattering (as is the case for transition metals)
- n=2 implies that the resistance is due to electron–electron interaction.
As the temperature of the metal is sufficiently reduced (so as to ‘freeze’ all the phonons), the resistivity usually reaches a constant value, known as the residual resistivity. This value depends not only on the type of metal, but on its purity and thermal history. The value of the residual resistivity of a metal is decided by its impurity concentration. Some materials lose all electrical resistivity at sufficiently low temperatures, due to an effect known as superconductivity.
An even better approximation of the temperature dependence of the resistivity of a semiconductor is given by the Steinhart–Hart equation:
where A, B and C are the so-called Steinhart–Hart coefficients.
This equation is used to calibrate thermistors.
In non-crystalline semi-conductors, conduction can occur by charges quantum tunnelling from one localised site to another. This is known as variable range hopping and has the characteristic form of 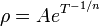 , where n=2,3,4 depending on the dimensionality of the system.
, where n=2,3,4 depending on the dimensionality of the system.
Complex resistivity and conductivity
When analyzing the response of materials to alternating electric fields, in applications such as electrical impedance tomography,[14] it is necessary to replace resistivity with a complex quantity called impeditivity (in analogy to electrical impedance). Impeditivity is the sum of a real component, the resistivity, and an imaginary component, the reactivity (in analogy to reactance). The magnitude of Impeditivity is the square root of sum of squares of magnitudes of resistivity and reactivity.
Conversely, in such cases the conductivity must be expressed as a complex number (or even as a matrix of complex numbers, in the case of anisotropic materials) called the admittivity. Admittivity is the sum of a real component called the conductivity and an imaginary component called the susceptivity.
An alternative description of the response to alternating currents uses a real (but frequency-dependent) conductivity, along with a real permittivity. The larger the conductivity is, the more quickly the alternating-current signal is absorbed by the material (i.e., the more opaque the material is). For details, see Mathematical descriptions of opacity.
Resistivity density products
In some applications where the weight of an item is very important resistivity density products are more important than absolute low resistivity- it is often possible to make the conductor thicker to make up for a higher resistivity; and then a low resistivity density product material (or equivalently a high conductance to density ratio) is desirable. For example, for long distance overhead power lines— aluminium is frequently used rather than copper because it is lighter for the same conductance.
| Material | Resistivity [nΩ·m] | Density [g/cm³] | Resistivity-density product [nΩ·m·g/cm³] |
|---|---|---|---|
| Sodium | 47.7 | 0.97 | 46 |
| Lithium | 92.8 | 0.53 | 49 |
| Calcium | 33.6 | 1.55 | 52 |
| Potassium | 72.0 | 0.89 | 64 |
| Aluminium | 28.20 | 2.70 | 72 |
| Copper | 16.78 | 8.96 | 150 |
| Silver | 15.87 | 10.49 | 166 |
| Gold | 22.14 | 19.30 | 427 |
| Iron | 96.1 | 7.874 | 757 |
Silver, although it is the least resistive metal known, has a high density and does poorly by this measure. Calcium and the alkali metals make for the best products, but are rarely used for conductors due to their high reactivity with water and oxygen. Aluminium is far more stable.
See also
- Conductivity near the percolation threshold
- Electric effective resistance
- Electrical resistivities of the elements (data page)
- Electrical resistivity imaging
- Ohm’s law
- Sheet resistance
- SI electromagnetism units
- Skin depth
- Superconductivity
Notes and references
- Notes
- ^ The numbers in this column increase or decrease the significand portion of the resistivity. For example, at 30 °C (303 K), the resistivity of silver is 1.65×10−8. This is calculated as Δρ = α ΔT ρo where ρo is the resistivity at 20 °C (in this case) and α is the temperature coefficient.
- ^ Referred to as 100% IACS or International Annealed Copper Standard. The unit for expressing the conductivity of nonmagnetic materials by testing using the eddy-current method. Generally used for temper and alloy verification of aluminium.
- ^ Gold is commonly used in electrical contacts because it does not easily corrode.
- ^ Commonly used for high voltage power lines
- ^ 18% chromium/ 8% nickel austenitic stainles steel
- ^ Nickel-Iron-Chromium alloy commonly used in heating elements.
- ^ Graphite is strongly anisotropic.
- ^ a b c The resistivity of semiconductors depends strongly on the presence of impurities in the material.
- ^ Corresponds to an average salinity of 35 g/kg at 20 °C.
- ^ This value range is typical of high quality drinking water and not an indicator of water quality
- ^ Conductivity is lowest with monoatomic gases present; changes to 1.2 × 10-4 upon complete de-gassing, or to 7.5 × 10-5 upon equilibration to the atmosphere due to dissolved CO2
- References
- ^ a b c d e f g h i j k l m n o Serway, Raymond A. (1998). Principles of Physics (2nd ed ed.). Fort Worth, Texas; London: Saunders College Pub. p. 602. ISBN 0-03-020457-7.
- ^ a b c d Griffiths, David (1999) [1981]. “7. Electrodynamics”. In Alison Reeves (ed.). Introduction to Electrodynamics (3rd edition ed.). Upper Saddle River, New Jersey: Prentice Hall. p. 286. ISBN 0-13-805326-X. OCLC 40251748.
- ^ http://physics.mipt.ru/S_III/t (PDF format; see page 2, table in the right lower corner)
- ^ a b Giancoli, Douglas C. (1995). Physics: Principles with Applications (4th ed ed.). London: Prentice Hall. ISBN 0-13-102153-2.
(see also Table of Resistivity) - ^ John O’Malley, Schaum’s outline of theory and problems of basic circuit analysis, p.19, McGraw-Hill Professional, 1992 ISBN 0070478244
- ^ Glenn Elert (ed.), “Resistivity of steel”, The Physics Factbook, retrieved and archived 16 June 2011.
- ^ Y. Pauleau, Péter B. Barna, P. B. Barna, Protective coatings and thin films: synthesis, characterization, and applications, p.215, Springer, 1997 ISBN 0792343808.
- ^ Hugh O. Pierson, Handbook of carbon, graphite, diamond, and fullerenes: properties, processing, and applications, p.61, William Andrew, 1993 ISBN 0815513399.
- ^ Lawrence S. Pan, Don R. Kania, Diamond: electronic properties and applications, p.140, Springer, 1994 ISBN 0792395247.
- ^ Physical properties of sea water
- ^ Pashley, R. M.; Rzechowicz, M; Pashley, LR; Francis, MJ (2005). “De-Gassed Water is a Better Cleaning Agent”. The Journal of Physical Chemistry B 109 (3): 1231. doi:10.1021/jp045975a. PMID 16851085.
- ^ Ohring, Milton (1995). Engineering materials science, Volume 1 (3rd edition ed.). p. 561.
- ^ Pawar, S. D.; Murugavel, P.; Lal, D. M. (2009). “Effect of relative humidity and sea level pressure on electrical conductivity of air over Indian Ocean”. Journal of Geophysical Research 114: D02205. Bibcode 2009JGRD..11402205P. doi:10.1029/2007JD009716.
- ^ [1]
Further reading
- Paul Tipler (2004). Physics for Scientists and Engineers: Electricity, Magnetism, Light, and Elementary Modern Physics (5th ed.). W. H. Freeman. ISBN 0-7167-0810-8.
External links
- New nanomaterial better efficient conductor
- Resistivity & Mobility Calculator/Graph from BYU cleanroom
- Bucknell University
This information originally retrieved from http://en.wikipedia.org/wiki/Electrical_conductivity
on Wednesday 3rd August 2011 9:49 pm EDT
Now edited and maintained by ManufacturingET.org